Kissei Pharmaceutical halts Amgen drug prescriptions after 20 patient deaths in Japan
Here's what it means for you.
This incident underscores critical concerns regarding drug safety and regulatory practices in the pharmaceutical sector.
What happened
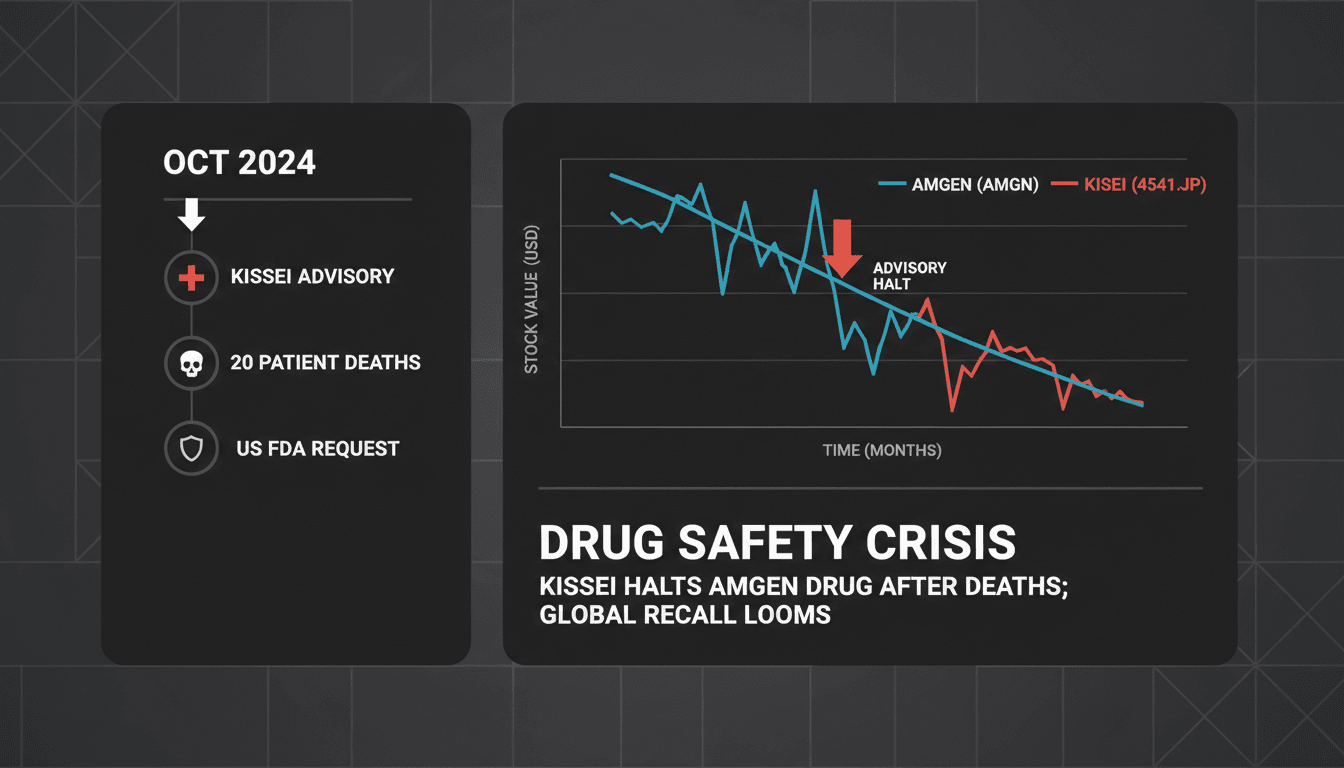
Twenty people in Japan died after taking an Amgen drug, leading to warnings from Kissei Pharmaceutical.
The Context
- Kissei Pharmaceutical is the distributor of the drug in Japan.
- U.S. regulators have intervened, asking for a voluntary withdrawal of the drug.
- Amgen's stock has fallen following the news of the deaths and warnings.
Takeaway
The incident raises significant questions about drug safety and regulatory oversight in the pharmaceutical industry.
This article was generated by AI from 3 verified sources and reviewed by A47 editorial systems.
U.S. business news, corporate developments, and economy.
"The Wall Street Journal is respected for deep financial and economic reporting with a center-right editorial perspective."
— A47 Editor
20 People in Japan Died After Taking Amgen Drug
In Japan, 20 individuals have died after taking the immune-disease drug Tavneos, marketed by Kissei Pharmaceutical, prompting the company to advise doctors against prescribing it to new patients. This alarming development has raised serious concerns ...
Public health, medical research, healthcare policy, and pharmaceutical developments.
"Dow Jones is a major financial news provider with a professional tone and broad coverage, including specialized health-sector reporting."
— A47 Editor
Twenty people in Japan who took an Amgen rare immune-disease drug have died, according to the company that sells the medicine in Japan. U.S. regulators had asked Amgen to voluntarily withdraw the drug.
Twenty individuals in Japan have died after taking a rare immune-disease drug sold by Kissei Pharmaceutical, prompting the company to advise doctors against prescribing it to new patients. This warning follows a request from U.S. regulators for Amgen...
Market-moving headlines impacting equities, bonds, and related risk assets.
"Real-time catalysts and volatility drivers across indices and sectors."
— A47 Editor
Amgen stock falls after Kissei warns on Tavneos liver risks
Amgen's stock has declined following a warning from Kissei regarding potential liver risks associated with its drug Tavneos. This announcement has raised concerns among investors about the safety profile of the medication, which could impact its mark...